 
SRP is composed of six proteins (P9/P14, P68/P72, P19, and P54) and a 300-nucleotide RNA scaffold. As protein synthesis begins, the signal sequence is recognized by a signal recognition particle 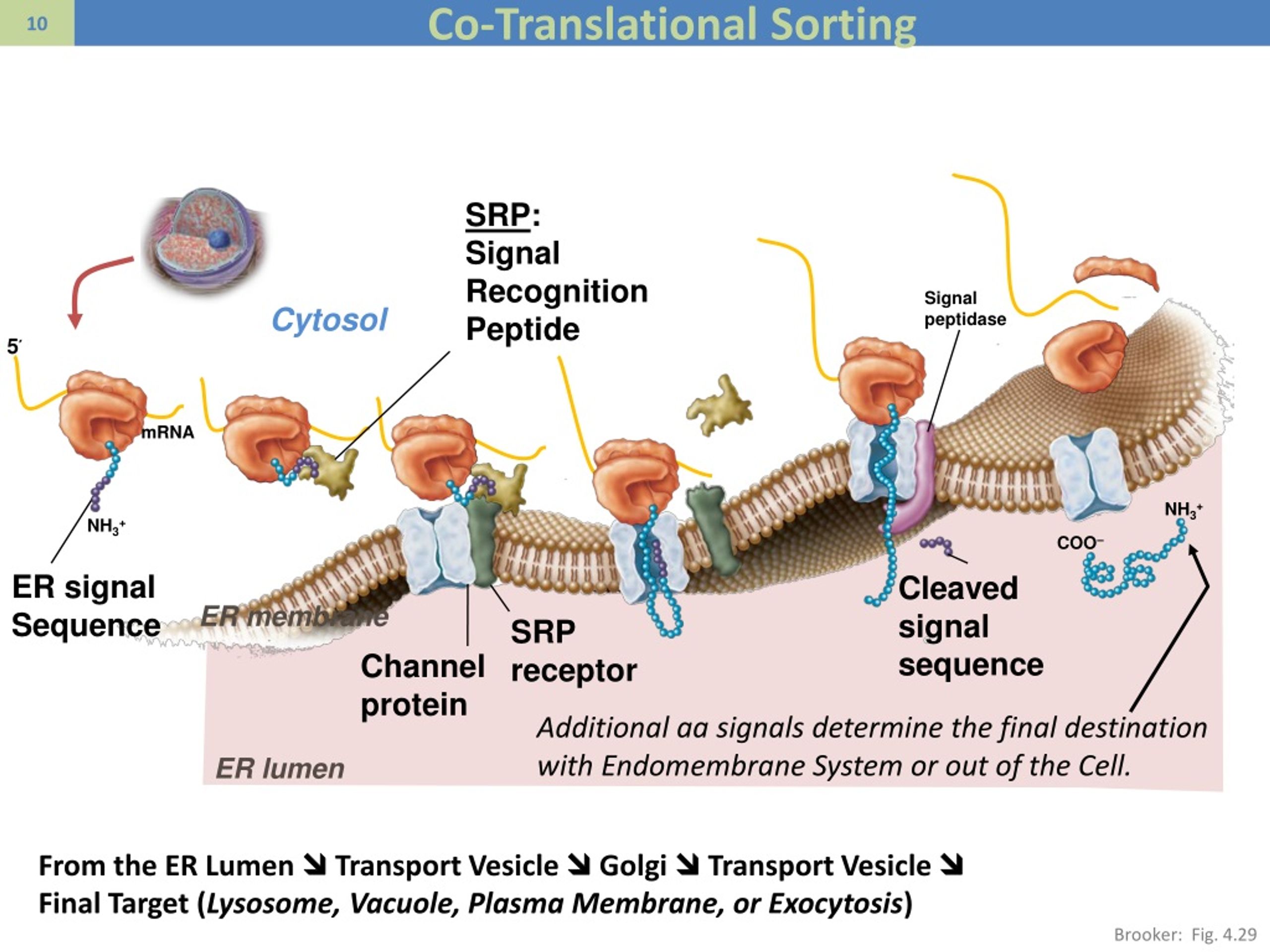
Signal sequences are quite variable, but generally have 6 to 12 hydrophobic amino acids flanked by one or more The cotranslational targeting of secreted proteins to the ER is achieved by an amino-terminal signal sequence of 16–30Īmino acids. Some proteins, particularly small polypeptides or proteins with “weak” signal sequences, can access the ER posttranslationally In eukaryotic cells, most secreted proteins are cotranslationally translocated from ribosomes into the rough ER. Previous Section Next Section TARGETING OF PROTEINS TO THE ER 
(S–S) Disulfide bond (□) N-Acetyl glucosamine (○) mannose (▵) glucose. Note that protein folding and disulfide bond formation happen rapidly after the nascent protein emergesįrom the translocon. Glycoproteins into ER exit sites followed by packaging into appropriate compartments, for example, the ER Golgi intermediateĬompartment (ERGIC). (5) Introduction/correction of disulfide bonds by protein disulfide isomerases/oxidoreductases. (4) Correct folding and quality control by (3) Addition of N-glycans to Asn residues of glycoproteins by oligosaccharyl transferase (OST). Of the (glyco)protein into the ER by the translocon. The signal recognition particle (SRP) and its receptor, which helps position the emerging protein at the translocon. (1) Correct targeting of the glycoprotein to the ER as it emerges from the translocon. Six key points in the fidelity of protein secretionĪre illustrated. Overview of protein targeting to the ER and glycoprotein quality control. Machinery, the provision of disulfide bonds, and the ER exit strategies used by proteins to ensure their efficient secretion Here, the focus is on recent advances in our understanding of the glycosylation Some proteins require assembly into complexes, and provision of lipids for ER membranes and the regulation of cholesterolĬontent are also important considerations. The folding and quality control of the protein, the availability of cofactors (particularly calcium), and the appropriateįormation of disulfide bonds ( Braakman and Bulleid 2011). Nascent protein to the ER by the ribosome, the translocation of the protein into the ER, the provision of sugars for glycoproteins, To ensure orderly ER exit, protein secretion from the ER is determined by several factors. Many of these proteins can also be alternatively spliced, adding a further layer of complexity ( Carninci et al. Or multi-pass transmembrane proteins that get targeted to the ER ( Kanapin et al. It has been calculated that the products of 11% of the approximatelyĢ5,000 predicted human full-length open reading frames (ORFs) are soluble secretory proteins, with a further 20% being single-pass Is a question of considerable importance in biology. How a cell controls its complex output and ensures that only properly folded and functional proteins reach their correct destination Proteins for secretion, with an emphasis on glycoprotein quality control and pathways of disulfide bond formation. This article describes recent advances in our understanding of the ER and its role in preparing However, there remain gaps in our knowledge, particularly when applying insight gained from model systems to the more complex Our understanding of how protein secretion in general is controlled in the ER is now quite sophisticated. Pancreas, producing insulin and other hormones, and mature B cells, producing antibodies, place a great demand on their endoplasmic Whereas some quiescent cell types secrete minor amounts of proteins, tissues like the In a complex multicellular organism, different cell types engage in specialist functions, and as a result, the secretory output 
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
